
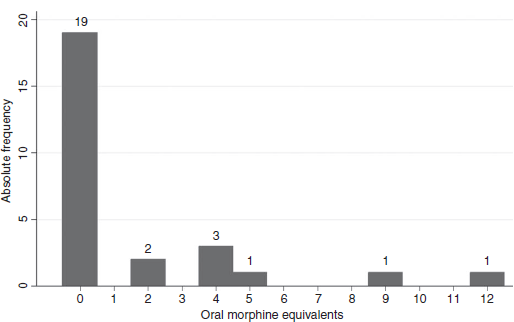
Postoperative analgesia, which will be assessed by total analgesic consumption, and time till administration of first rescue analgesia will be recorded.Post-operative pain will be assessed by Numeric Rating Scale (NRS) from 0 to 10 at 0,2,4,6 then every 6h for 24 hours and in the second and third postoperative days at rest and during Physiotherapy.
IPACK BLOCK CPT CODE PLUS
All patients will receive spinal anesthesia with 3 ml 0.5% (15 mg) hyperbaric bupivacaine plus 25 μgm fentanyl at the 元/4 interspaces and the study of regional anesthetic technique will be performed postoperatively according to each group. On entering the operating room, intravenous line (IV) will be inserted, routine monitoring of heart rate by ECG, noninvasive blood pressure (NIBP), pulse oximetry will be done. During the pre-anesthetic assessment, all patients will be familiarized with Numeric Rating Scale (NRS) score.
History talking, clinical examination, routine laboratory investigations including CBC, coagulation profile, random blood sugar, liver and renal function tests. Group (III): 20 patients will receive adductor canal block (ACB) and infiltration of the interspace between popliteal artery and the capsule of posterior knee block (IPACK). Group (II): 20 Patients will receive peri-articular injection alone. Group (I): 20 patients will receive adductor canal block alone. Randomization will be done by computer generated numbers into:
IPACK BLOCK CPT CODE CODE
Every patient will receive an explanation to the purpose of the study and every patient will have a secret code number to ensure privacy to participants and confidentiality of data.Īdult patients with severe osteoarthritis, ASA class I, II and III and scheduled for elective Total knee arthroplasty will be enrolled in the study. Any unexpected risks encountered during the course of the research will be cleared to the participants as well as to the Ethical Committee on time. All data of the patients will be confidential with secret codes and private file for each patient, all given data will be used for the current medical research only. After approval from institutional ethics committee, an informed consent will be taken from each patient. This prospective randomized study will be carried out in Tanta University Hospitals in Anesthesia Department for one year after approval from ethical committee of faculty of medicine of Tanta university. Why Should I Register and Submit Results?.
